
Non-BCMA targeting bispecific antibodies are a relatively new class of therapeutics. In our Patient Webinar on October 11, 2023, Drs. Ajai Chari (UCSF) and Suzanne Trudel (University of Toronto, Princess Margaret Hospital, Ontario, Canada) provided fascinating updates on this emerging class of agents.
Bispecific antibodies belong to the drug class known as T-cell engagers, which also includes CAR T-cells. Unlike CAR T-cells, which are engineered (over the course of 4-6 weeks) from a patient’s own T-cells to better recognize and kill myeloma cells, bispecific antibodies are an “off-the-shelf” treatment that can bind to both myeloma cells and to T cells at the same time, bringing them in close proximity so that the T cells can recognize and kill the myeloma cells. There are now three bispecific antibody therapies approved in the US for myeloma patients who have received at least four previous lines of therapy; two of these bind to BCMA on the surface of myeloma cells, and one binds to a molecule called GPRC5D on the surface of myeloma cells.
Dr. Chari began by discussing Talvey (talquetamab), a GPRC5D-targeting bispecific antibody approved for use in the US. Historically speaking, the bispecific antibody drug class offers increased activity in highly pre-treated myeloma patients (those who have already received 4 or more lines of therapy). The relative activity of a number of different drugs in this patient population is seen in this slide:
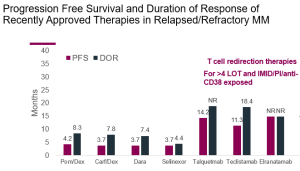
In comparison to current standards of care, such as Pomalyst/dexamethasone, it is clear from this figure that the three currently approved bispecifics—teclistamab (Tecvayli), talquetamab (Talvey), and elranatamab (Elrexfio)—have superior activity in this patient population in terms of both progression-free survival and duration of response (NR means the duration of response was not reached, as so many patients were still responding at the time the data was collected). Dr. Chari also mentioned that of the 288 patients enrolled in the Talvey Phase1/2 trials, 51 had received prior therapy with T-cell engagers, and their response rate compared to those who had not previously received such therapy was only slightly lower (65% vs 73%).
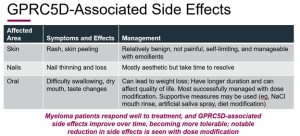
Regarding side effects and adverse occurrences, Dr. Chari provided some further fascinating facts on Talvey not demonstrated on a slide. Dr. Chari stated that patients who experienced more severe side effects (particularly skin rashes and nail issues) also responded better to Talvey. As a result, there is a correlation between side effects and pharmacological action, which may give patients who are experiencing discomfort from side effects optimism that the medication may work for them. He added that patients with the most severe adverse effects can benefit from other treatments or dose decrease, and that generally, these side effects do get better with time.
Next, Dr. Trudel discussed cevostamab, a FcRH5-targeting bispecific antibody that is still in clinical trials. Two noteworthy pieces of information emerged from her presentation:
Thank you to these two amazing doctors, and also to our patient speaker, Nick Lenoir, for sharing their knowledge! Nick was especially interesting, as he is currently receiving treatment with Talvey and is having issues with side effects, so he was very happy to hear that this meant he is more apt to respond to this therapy.
For more information on bispecific antibodies and other myeloma therapies, please visit our Treatment Options page https://themmrf.org/diagnosis-and-treatment/treatment-options/.